 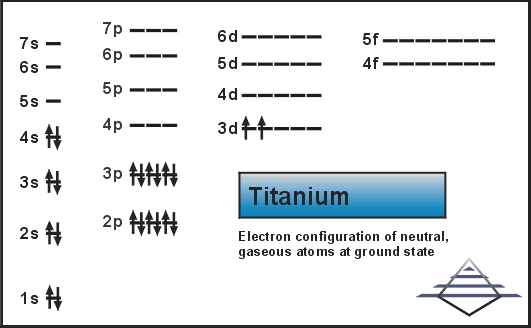
However, there is an exception for the d-block and f-block, in which the energy level, 'n' for the d block is. The energy level, 'n', can be determined based on the periodic table, simply by looking at the row number in which the element is in. Here # represents the electron configuration of argon, the noble gas that comes immediately before titanium in the periodic table. In the case of first row transition metals, the electron configuration would simply be Ar 4s x 3d x. Namely, the observed change of the valence-electron configurations of metals in alloys can be explained with the transfer of 3d electrons from one element to. If you want, you can use the noble gas shorthand notation to write The electronic configuration of Ti is Ar 3d 4s. Therefore, the two electrons that are lost when the #"Ti"^(2+)# is formed will come from the #4s# orbital, which means that the electron configuration of the cation is Titanium (Ti) is a transition element with the atomic number 22. 
This means that when titanium loses electrons, it does so from the #4s# orbital first. However, once the #4s# orbital is filled, it becomes higher in energy than the #3d# orbitals. That happens because the empty #3d# orbitals are actually higher in energy than the empty #4s# orbital, as seen here Now, it's important to keep in mind that this notation for the electron configuration is useful when adding electrons to build an atom "from scratch" because in that case, the #4s# orbital is filled before the #3d# orbitals. The electron configuration of a neutral titanium atom looks like this Consequently, the electron configuration of the titanium(II) cation, #"Ti"^(2+)#, must account for #20# electrons, since this cation is formed when a neutral titanium atom loses #2# electrons. Therefore, the electron configuration of a neutral titanium atom must account for #22# electrons. This means that a neutral titanium atom will contain #22# protons in its nucleus and #22# electrons surrounding its nucleus. 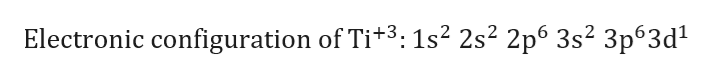
In this case, titanium, #"Ti"#, is located in period 4, group 4 of the periodic table and has an atomic number of #22#. A good place to start when trying to figure out the electron configuration of an ion is the electron configuration of the neutral parent atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
